Let us pause for a moment and consider the structure of the atom as we know it now. Every atom is made from three kinds of elementary particles: protons, which have a positive electrical charge; electrons, which have a negative electrical charge; and neutrons, which have no charge. Protons and neutrons are packed into the nucleus, while electrons spin around outside. The number of protons is what gives an atom its chemical identity. An atom with one proton is an atom of hydrogen, one with two protons is helium, with three protons is lithium, and so on up the scale. Each time you add a proton you get a new element. (Because the number of protons in an atom is always balanced by an equal number of electrons, you will sometimes see it written that it is the number of electrons that defines an element; it comes to the same thing. The way it was explained to me is that protons give an atom its identity, electrons its personality.)
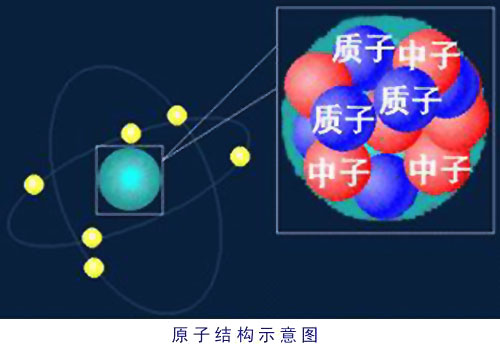
Neutrons don't influence an atom's identity, but they do add to its mass. The number of neutrons is generally about the same as the number of protons, but they can vary up and down slightly. Add a neutron or two and you get an isotope. The terms you hear in reference to dating techniques in archeology refer to isotopes—carbon-14, for instance, which is an atomof carbon with six protons and eight neutrons (the fourteen being the sum of the two).











